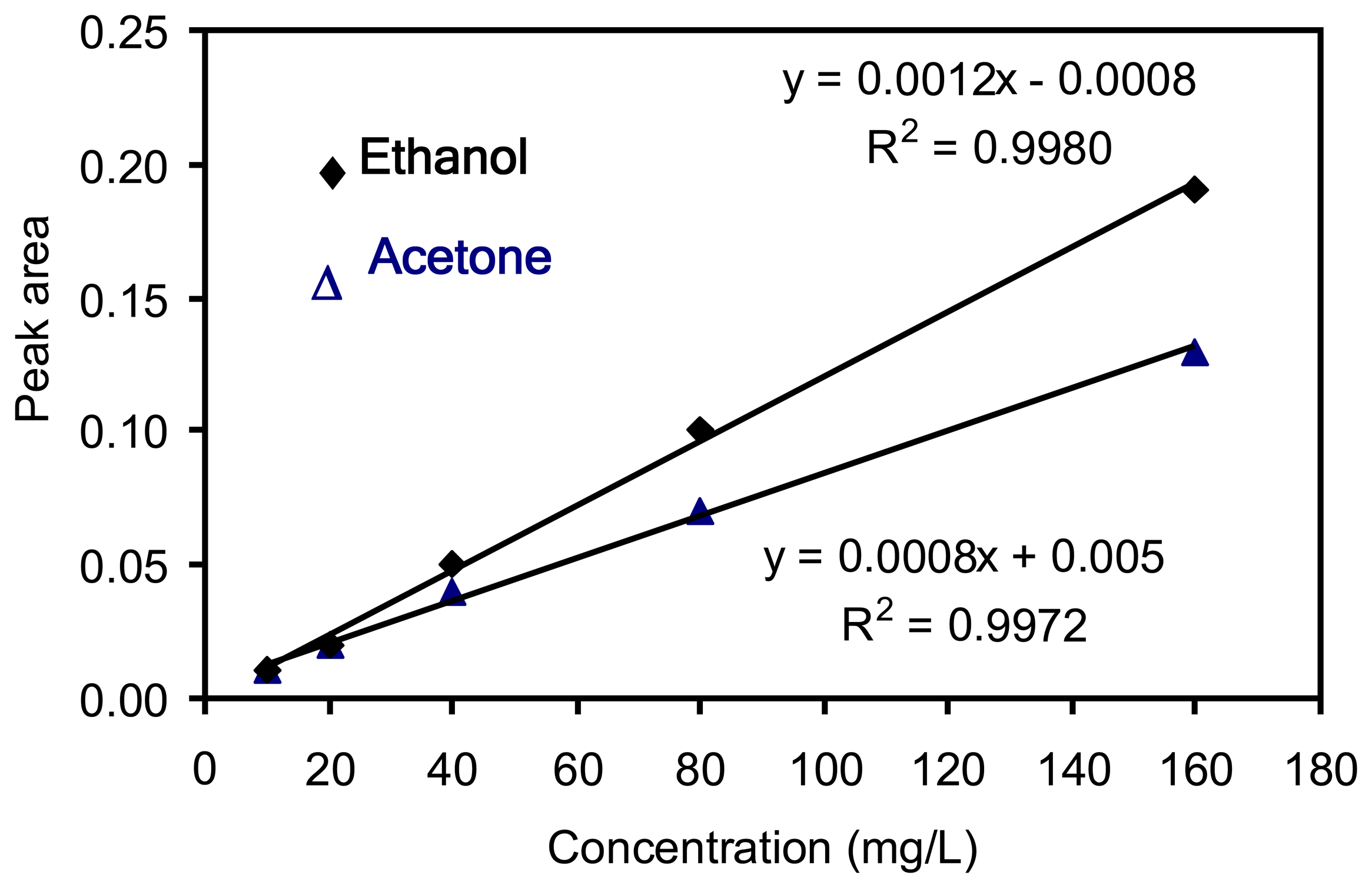
Chemicals from ethanol: the acetone synthesis from ethanol employing Ce0.75Zr0.25O2, ZrO2 and Cu/ZnO/Al2O3 | BMC Chemistry | Full Text
![PDF] Separation of butanol from acetone-butanol-ethanol fermentation by a hybrid extraction-distillation process | Semantic Scholar PDF] Separation of butanol from acetone-butanol-ethanol fermentation by a hybrid extraction-distillation process | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/488fa67db9c5b3e56955a272ff8794a9953afdf7/4-Table1-1.png)
PDF] Separation of butanol from acetone-butanol-ethanol fermentation by a hybrid extraction-distillation process | Semantic Scholar

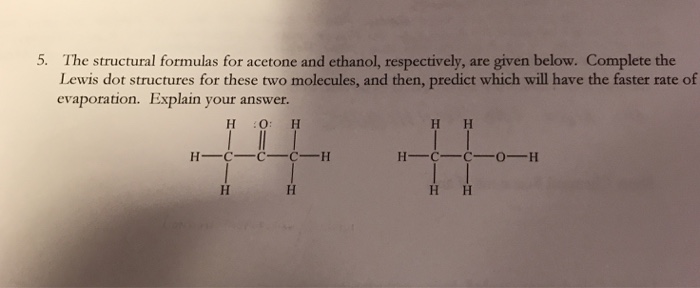
Explain the reaction between acetone and ethanol - Chemistry - Solutions - 12698165 | Meritnation.com
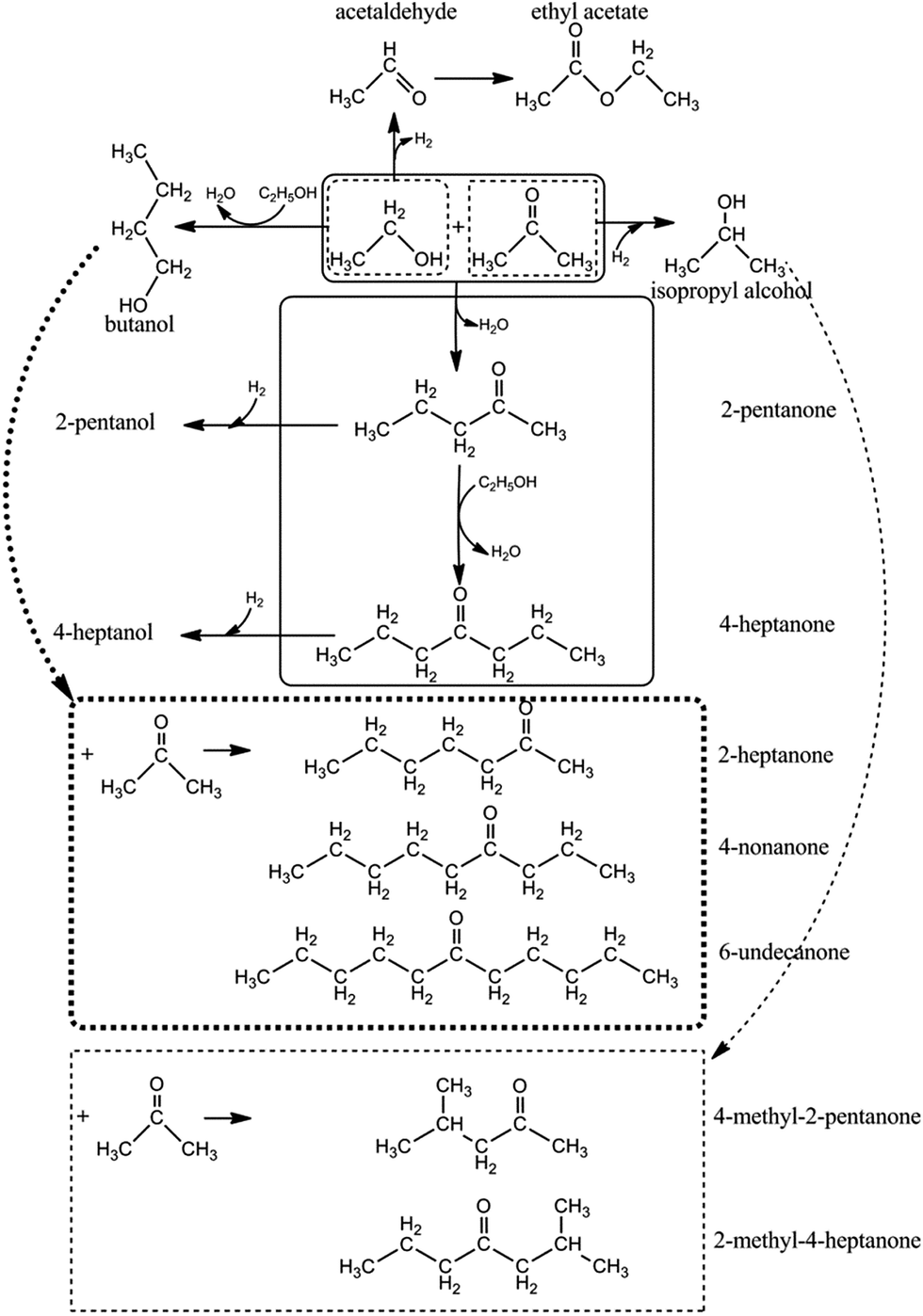
Acetone alkylation with ethanol over multifunctional catalysts by a borrowing hydrogen strategy - RSC Advances (RSC Publishing) DOI:10.1039/C5RA17889D

Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is:

Structure formula and size of acetone, ethanol, and water, considering... | Download Scientific Diagram
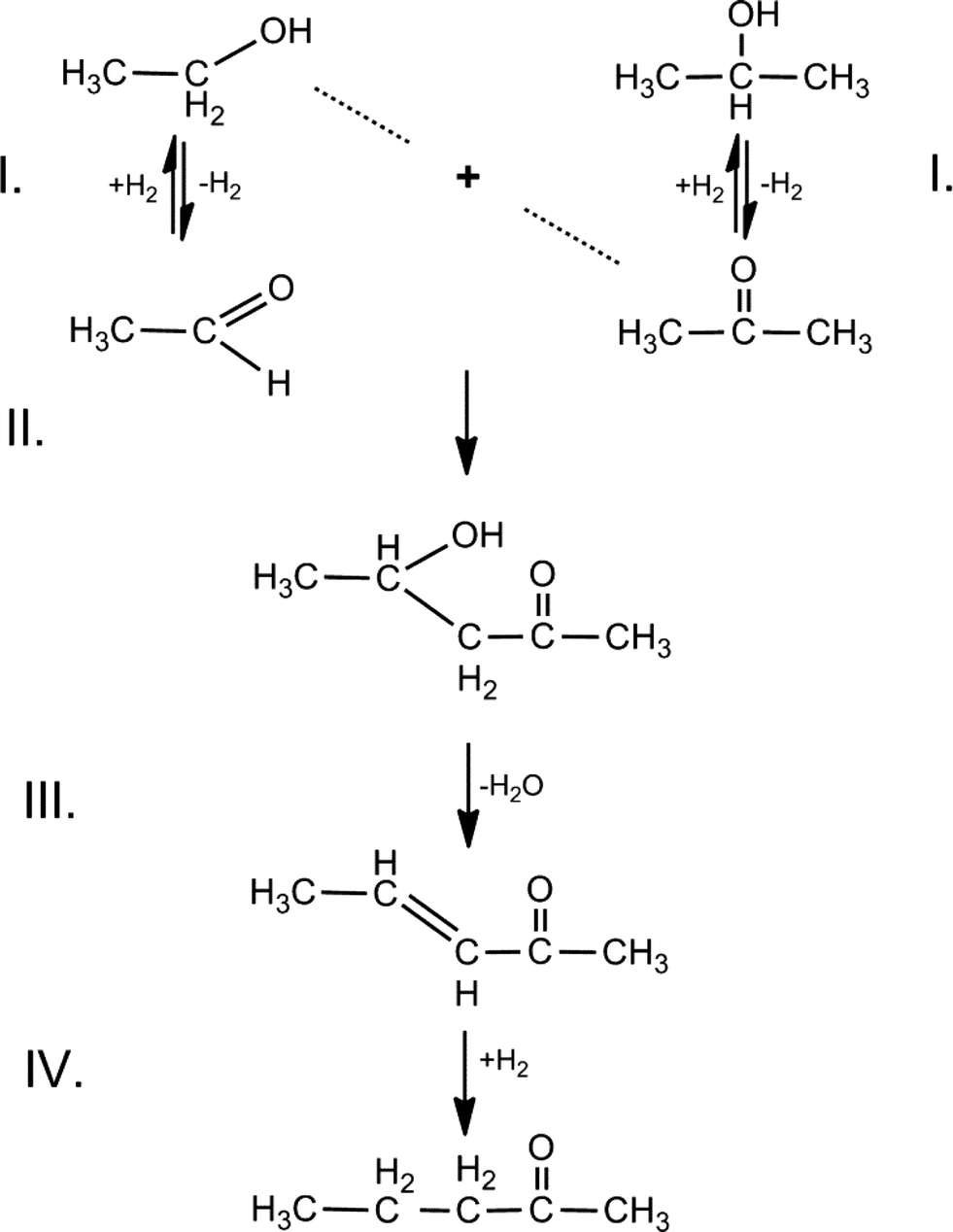
Upgraded biofuel from alcohol–acetone feedstocks over a two-stage flow-through catalytic system - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C6CY00025H

solutions - What are the intermolecular forces between the following compounds in a mixture? - Chemistry Stack Exchange

Draw the structures of ethanol, acetone, toluene, hexane, and water. Classify each solvent as polar, nonpolar, or moderately polar. | Homework.Study.com

thermodynamics - What mixing ratio of ethanol and acetone has the lowest freezing point? - Chemistry Stack Exchange